Ibuprofen – C13H18O2 – 206.285
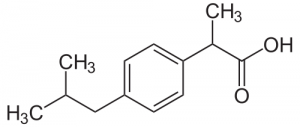
Structural Formula (1)
Ibuprofen is one of the most popular NSAID (Non-steroidal Anti-inflammataory Drug) Drugs, used to treat pain, fevers, and inflammation. It comes in several forms, but we are focusing on the tablets. Ibuprofen is derived from 2-methylpropylbenzene. 2-methylpropylbenzene is made from compounds that are separated from crude oils and has the chemical formula C10H13+. Ibuprofen is used to block the production of prostaglandins. These are chemicals that the body produces when someone is sick or injured. They make nearby nerves sensitive to pain, so your body can react and start the healing process. They can also swell tissue depending on the injury/sickness.
Design
The design of Ibuprofen is two 2-methylpropylbenzene put together. There are 3 de localized pi orbitals. The negative region of the medicine is where it bonds with oxygen. The positive side is the side where Oxygen bonds with Hydrogen[specifically the Hydrogen]. The C-C bonds are the longest bonds because Carbon is the largest element in the molecule, and it is also the weakest because of the length of the bond.

3D Model of Ibuprofen (2)
The O-H bond is the shortest bond because Oxygen and Hydrogen are two smallest elements in the molecule. The strongest bond is the C=O bond because it is a double [pi] bond, which is stronger than single [sigma] bonds.

Delivery

Ibuprofen Pill (3)
Ibuprofen is taken orally in the form of a pill. It is generally advised to be taken after consuming food. In order for it to take effect, ibuprofen must first travel through the body. It goes down the esophagus, eventually residing in the stomach. Since Ibuprofen is not polar (seen in the design page), it can safely travel through the polar bloodstream without being dissolved. The outside of the pill allows the fragile inside to safely through the stomach without being dissolved early. Once there, the gastric acid dissolves it. This allows the ibuprofen to be absorbed into the bloodstream through the intestinal wall, and go to the area that is releasing the prostaglandinas. Over 98% of the Ibuprofen is bound to plasma proteins, and it can also get access to the central nervous system. Ibuprofen will travel to almost anywhere in the body if needed, because of the high variability of where prostaglandinas can be produced. It works by latching onto to an enzyme called cyclooxygenase, blocking it out, which stops it from making the prostaglandins.
Destruction

Dissolved Ibuprofen (3)
The lifetime of Ibuprofen in the body depends on the person consuming it. Usually the drug will be fully absorbed within 30 minutes to 2 hours after taken orally. The Half life of the drug varies between 1.8 to 2 hours. When it comes time for the drug to leave the body, it is excreted as urine. More than 90% of the drug is excreted through urine as metabolites (a substance necessary for metabolism).
2C13H18O2 + 33O2 —> 26CO2 + 18H2O
1gram C13H18O2 * 1mol C13H18O2 / 206.285grams C13H18O2 = 0.0048476622mol C13H18O2
0.0048476622mol C13H18O2 / 2mol C13H18O2 * 33mol O2 = 0.0799864265mol O2
0.0799864265mol O2 * 32grams O2 / 1mol O2 = 2.559565648grams O2 required to oxidize 1gram of Ibuprofen
0.0048476622mol C13H18O2 / 2mol C13H18O2 * 26mol CO2 = 0.0630296086mol CO2
0.0630296086mol CO2 * 44grams CO2 / 1mol CO2 = 2.772862778grams CO2 produced from 1gram of Ibuprofen
0.0048476622mol C13H18O2 / 2mol C13H18O2 * 18mol H2O = 0.0436289598mol H2O
0.0436289598mol H2O * 18grams H2O / 1mol H2O = 0.7853212764grams H2O produced from 1gram of Ibuprofen
After 24 hours the drug is completely excreted from the body, a majority not changed from the form it entered.
Image Sources
- https://chemistry.stackexchange.com/questions/63308/naming-ibuprofen
- https://commons.wikimedia.org/wiki/File:(S)-ibuprofen-3D-balls.png
- https://www.webmd.com/drugs/2/drug-5166-9368/ibuprofen-oral/ibuprofen-oral/details
- Victor Urnyshev


In the drawing, the leftmost carbon that was highlighted blue to indicated that it was a chiral carbon is incorrect. There is only one chiral carbon in ibuprofen, as you can see that carbon has two of the came CH3 groups, making in not chiral.
The carbon circled in blue on the left is not chiral – it has two of the same groups (methyls).